Introduction
Titanium bone screws turning is the precision CNC turning (often on Swiss-type lathes) used to produce implant-grade screw geometries, including the shank, thread, and driver features, from titanium bar stock. The parts are small, but the consequences of variation are large. A minor burr at the thread crest, a rounded driver corner, or a traceability gap can become a quality hold, a delayed build, or a regulatory headache.
Grade 23 titanium, also called Ti-6Al-4V ELI (extra low interstitial), matters because it is widely specified for surgical implant applications where toughness and fatigue performance are critical and where material chemistry limits are tighter than standard Ti-6Al-4V. In practical sourcing terms, Grade 23 raises the bar on incoming material control, lot traceability, contamination control, and process stability.
For engineers and sourcing teams, the question is rarely “can you machine titanium?” If you need a baseline for how CNC turning programs are typically structured, AFI’s overview of CNC Turning Services is a good internal reference for terminology and scope. It is whether a CNC turning partner can repeatedly hit geometry, surface condition, and documentation expectations across prototypes, verification builds, and production lots.
What you should evaluate:
- Material controls (Grade 23 requirements, MTR/MTR traceability, and standards alignment)
- DFM decisions that determine manufacturability and risk (threads, driver features, GD&T)
- CNC turning process controls (tooling strategy, chip and burr control, in-process measurement, SPC)
- Surface finish, cleaning validation, and packaging readiness
- Test capability and documentation discipline for regulatory-facing programs
Outcome: reduce risk, ensure compliance alignment, and accelerate NPI (new product introduction) without rework loops.
Key Takeaway: The fastest way to de-risk titanium grade 23 bone screw CNC turning is to treat it as a controlled system: material + geometry + process capability + cleanliness + records. If one is weak, the program slows down.
Table of Contents
Material & Standards Baseline
Ti-6Al-4V ELI requirements
Ti-6Al-4V ELI Grade 23 (also called Grade 23) is specified when the program needs implant-grade titanium with tighter limits on interstitial elements (such as oxygen, nitrogen, carbon, and iron) compared with standard Ti-6Al-4V. Those tighter limits are not just “paper requirements.” They affect ductility, fracture toughness, and fatigue behavior, and they change how conservative you should be about heat input, tool wear, and surface condition.
From a turning partner perspective, Grade 23 requirements usually translate to three shop-floor realities:
- Incoming material verification is non-negotiable. The supplier should be able to map bar heat/lot to every finished screw lot.
- Heat management and tool condition matter more than on free-machining alloys. Titanium’s low thermal conductivity concentrates heat at the tool–chip interface, so process drift can show up first as burr growth, smeared surfaces, or dimensional shift.
- Surface contamination control must be designed in. Chips, embedded foreign material, and residues can undermine downstream passivation/cleaning validation and create investigation work.
Traceability and MTRs
Medical programs live and die on traceability. At minimum, you should expect:
- Material test reports (MTRs) (often also called mill test certificates) that tie the bar stock heat/lot to chemistry and mechanical properties.
- Lot-to-lot traceability across receiving, cutting, turning, finishing, cleaning, and packaging.
- Identification controls so mixed lots do not happen during kitting, machine loading, or secondary operations.
A practical check: ask how the supplier prevents a “paper match” (documents present) from becoming a “physical mismatch” (parts and paperwork separated). The answer should include traveler control, barcode/label discipline, and where the traceability breakpoints are (cut lengths, WIP bins, outside processing).
Applicable ASTM/ISO references
The exact standards set depend on your design, intended use, and regulatory strategy, but a CNC turning partner should be fluent in the vocabulary and be able to build an inspection and documentation plan around it.
Common references you’ll see for Grade 23 titanium bone screws turning include:
- Material baseline standards for implant titanium, frequently referenced in FDA-facing contexts (for example, the FDA guidance document that lists consensus standards for orthopedic bone screws and materials, including ASTM F136 (wrought Ti-6Al-4V ELI) in the FDA’s orthopedic bone screw guidance PDF).
- Packaging standards for sterile barrier systems when the program includes terminal sterilization, such as ISO 11607-1:2019 (packaging for terminally sterilized medical devices).
- Cleanliness standards for orthopedic implants, such as the ISO 19227 (cleanliness of orthopedic implants) project page.
You don’t need a machine shop to “certify” standards that they don’t own. You do need them to understand what evidence your program will need, and to avoid process choices that make compliance harder later.
If your sourcing team also supports adjacent titanium programs, AFI’s page on Grade 5 Titanium Alloy CNC Machining can help align internal terminology between Grade 5 and Ti-6Al-4V ELI Grade 23 discussions.
DFM for Bone Screws
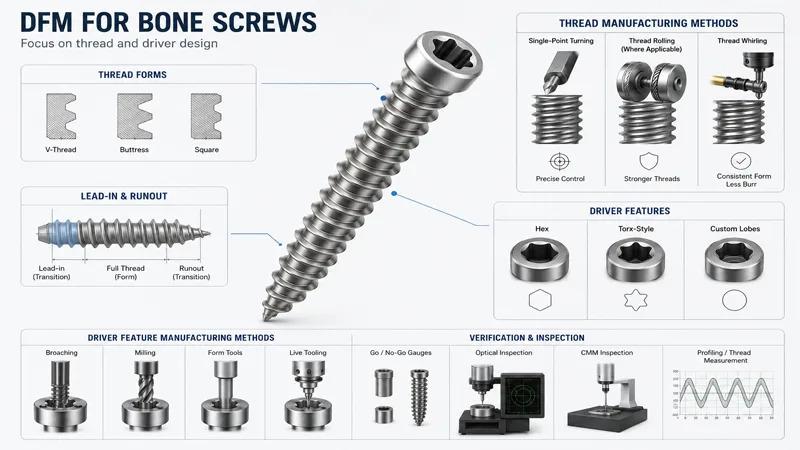
Thread forms and driver features
DFM for bone screws is about controlling risk where geometry meets function: threads, lead-in, and driver engagement.
Key decisions to settle early:
- Thread type and how it will be produced: single-point turning, thread rolling (where applicable), or thread whirling. For many medical screw geometries, whirling can reduce burr risk and keep thread form consistent by producing the thread in a controlled, continuous cut.
- Lead-in and runout strategy: where the full thread form starts and ends, and how you manage a transition zone without creating a stress riser or a burr trap.
- Driver feature geometry (hex, Torx-style, custom lobes): corners and depth matter. A driver that “almost fits” is a field failure waiting to happen, and it is also a metrology headache if no gauge strategy exists.
A supplier worth shortlisting can explain how driver features are made (broaching, milling, form tools, live tooling) and what they use to verify them beyond “it looks good.”
Tolerances and GD&T focus
Bone screws are often dimensionally small, but they are not simple. Typical tolerance risk clusters include:
- Concentricity/coaxiality between the shank, thread axis, and head features
- Major/minor diameters and pitch diameter (thread functional fit)
- Head-to-shank transitions, chamfers, and fillets (burr and fatigue risk)
- Runout on critical diameters that influence insertion torque and perceived “wobble.”
Ask where the supplier expects to hold “as-machined” capability and where they recommend design or tolerance changes. Good DFM feedback is not generic. It is tied to features: tool access, deflection, inspection method, and expected process capability.
If your drawing uses GD&T, the partner should be able to talk about (and you can sanity-check the tolerance language against AFI’s internal overview of Tight Tolerance CNC Machining):
- Datum strategy and whether it matches how the part will be fixtured/held
- How they will measure (gage pin, optical comparator, CMM, vision system) and what the measurement uncertainty looks like
- How will they manage gauge R&R (repeatability and reproducibility) on the measurement plan
Size matrix and worst-case builds
Bone screw programs frequently include a size family (diameter/length combinations). The risk is that “one size fits all” but the family does not.
A practical DFM approach:
- Build a size matrix early (diameter × length × thread pitch × driver type).
- Identify the worst-case combinations for machining and inspection (longest L/D ratio, smallest driver corners, tightest pitch diameter tolerance).
- Confirm whether the same process can cover the family or whether the supplier expects tooling/program forks.
Pro Tip: If you can only afford to validate a subset in early NPI, pick the geometry that is hardest to machine and hardest to inspect. If that one holds, the rest are usually easier.
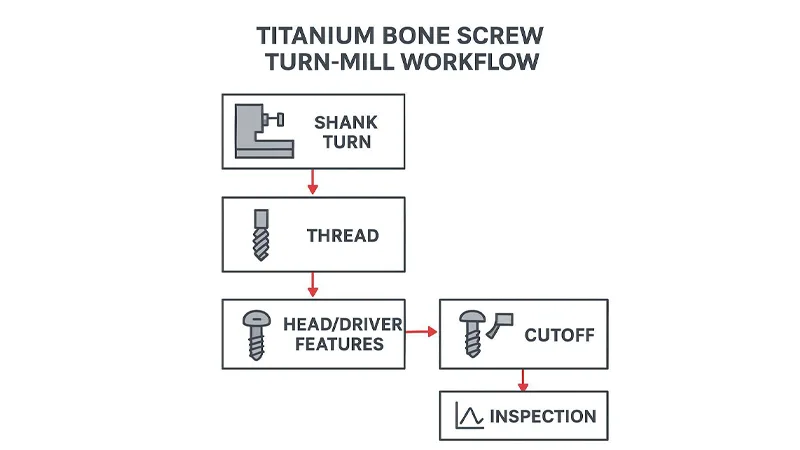
CNC Turning Process Controls (titanium grade 23 bone screw cnc turning)
Machines, tooling, and parameters
For titanium bone screws turning, the machine choice matters less than the control system around it. Swiss-type CNC lathes are common, and the broader category is often referred to as Swiss turning, for small diameters because the guide bushing supports the bar close to the cutting point, improving stability on long, slender parts.
Evaluate the supplier on (for general context on integrated operations, see CNC Turning and Milling Compound Machining):
- Machine configuration: sliding headstock (Swiss), sub-spindle transfer, live tooling capability, and whether the process is turn-only or turn-mill.
- Tooling plan: insert grades suitable for titanium, edge prep strategy for finishing cuts, and tool-life control.
- Parameter discipline: how cutting speed, feed, and depth of cut are locked down, and what triggers a parameter review.
Titanium’s heat behavior punishes “tweaking by feel.” A strong supplier treats parameters as part of the controlled process: documented, versioned, and linked to quality outcomes.
Coolant strategy and chip/burr control
Chip and burr control is a primary yield driver in titanium screw turning.
What to ask:
- Are they using non-chlorinated coolant strategies appropriate for titanium programs?
- Do they apply directed coolant or high-pressure coolant for chip breaking where needed?
- What is their method to prevent chips from recutting and marking the thread flanks?
- Where do burrs show up in their process (thread runout, driver pockets, cutoff) and what is the standard countermeasure?
Warning signs:
- “We’ll deburr it later” without specifying method and validation.
- Manual deburring as the default for production lots.
You want a process where burr minimization is designed into toolpath and tooling, and deburring is treated as a controlled step with acceptance criteria.
In-process SPC and measurement
In-process measurement is how the supplier catches drift before it becomes a batch problem.
A practical SPC (statistical process control) plan for titanium bone screws turning often includes:
- Tool-wear driven dimensions (major diameter, pitch diameter proxies)
- Critical head/driver dimensions that affect assembly and insertion
- Surface finish indicators where the program cares about Ra and defect-free flanks
Ask to see:
- Which characteristics are on control charts
- Sampling plan by build phase (prototype vs PV vs production)
- Reaction plan (what happens when the process trends)
Also check the measurement stack:
- How thread form is verified (functional gages vs optical methods)
- Whether measurement is automated/inline or bench-based
- How data is recorded and linked to lot/serial traceability
Surface Finish & Cleanliness
Finish targets and deburring
Surface finish is not just aesthetics. On screw threads, finish interacts with insertion torque, galling risk, and how the screw behaves during drive.
What “good” looks like depends on your design and surgeon-use scenario, but the supplier should be able to:
- State realistic finish targets by feature (thread flank vs shank vs head underside)
- Define deburring acceptance (no loose particles, no sharp edges that fail handling criteria)
- Explain the chosen deburring method and why it is stable for the geometry
For titanium, it’s common to treat burr prevention as a machining requirement (tool condition, cutting direction at exits, thread strategy) rather than a downstream cosmetic cleanup.
Passivation/anodizing choices
For implant programs, surface treatment decisions should be grounded in your design inputs and validation plan. Two common paths you may see:
- Passivation / surface preparation steps aligned to implant surface preparation practices (often structured around cleaning + acid treatment steps) to remove contaminants and embedded foreign matter.
- Titanium anodizing for identification color or surface property tuning, when specified by the program.
Important: do not let the supplier apply a “stainless steel passivation standard” by habit. Titanium surface preparation has its own accepted practices and must be validated against your cleanliness and biocompatibility requirements.
If anodizing is used for identification, confirm:
- Dimensional impact (oxide thickness control)
- Color consistency across lots
- Cleaning compatibility before and after anodizing
For general surface treatment context, AFI’s overview of anodizing options in anodized metals can be a useful internal reference for how anodizing options are typically described to non-specialist stakeholders.
Validated cleaning and packaging
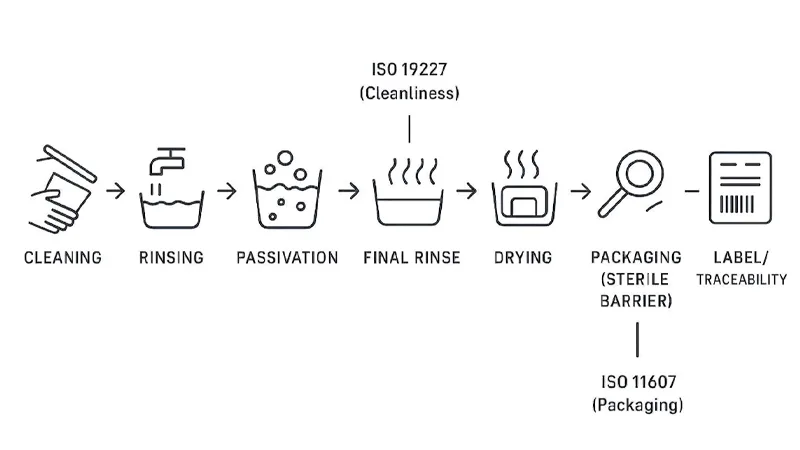
Cleanliness and packaging are where many machining suppliers get exposed. Even if your machining partner does not own final sterilization, their processes must not make validated cleaning impossible.
A sensible validation-oriented approach:
- Define the contamination risks upstream (coolant residues, chips, media, handling contamination).
- Validate cleaning with methods appropriate for orthopedic implant cleanliness targets and documentation expectations.
- Treat packaging as a system: material, forming/sealing process, and integrity testing.
ISO language is helpful for aligning teams:
- ISO cleanliness expectations are often framed using ISO 19227 (cleanliness of orthopedic implants).
- Packaging for terminally sterilized devices is framed under ISO 11607-1:2019, with validation of forming/sealing processes covered in the companion part of the standard series.
Testing, Quality, and Regulatory
ASTM F543 performance tests
Bone screw performance testing is often structured around standardized methods for torsion, driving characteristics, and fatigue behavior.
ASTM F543 is commonly referenced for metallic bone screw testing. It covers multiple test types (for example, torsional properties and fatigue-related methods) and provides a shared vocabulary across OEMs, labs, and reviewers.
A practical starting point is to confirm that your supplier understands what the tests represent and what the upstream manufacturing variables are that typically affect outcomes. Even if testing is performed at an outside lab, machining and finishing choices influence results.
For a concise overview of typical test setups and what is included, see ZwickRoell’s explainer on testing of bone screws to ASTM F543 and ISO 6475.
ISO 13485 documentation set
ISO 13485 is the quality management system (QMS) standard most often associated with medical device manufacturing and supply chains. Whether your machining partner is certified or operating under a customer-controlled QMS, the documentation behavior matters.
Expect an audit-friendly documentation set aligned to your build phase:
- Drawing and revision control
- Process flow and control plan (what is controlled, how, and with what reaction plan)
- Inspection plan and measurement records
- Nonconformance handling and corrective action trail when needed
- Traceability records linking material → WIP → finished lot
The point is not to drown in paperwork. It is to ensure that when a deviation happens (and at some point it will), the investigation can close quickly with defensible evidence.
U.S. 510(k) readiness and DHF/DHR
If the program is headed toward U.S. clearance, manufacturing records usually need to support the design history file (DHF) and device history record (DHR) expectations. Your machining partner should understand how their outputs feed that system.
Practical checks:
- Can they provide controlled build documentation on demand, tied to drawing rev and lot?
- Do they understand first article inspection (FAI) expectations and how to package those results?
- Can they support traceability in a way that survives handoffs (outside processing, cleaning, packaging)?
AFI Industrial Co., Ltd. supports DFM, SPC, FAI/PPAP deliverables, and lot traceability for precision turned parts in a documentation-first workflow.
⚠️ Warning: Regulatory readiness often fails in the seams: an outside-process step with weak traceability, a measurement method with poor repeatability, or a cleaning change that wasn’t revalidated.
Risks, Cost, and Scalability
Common failure modes and mitigations
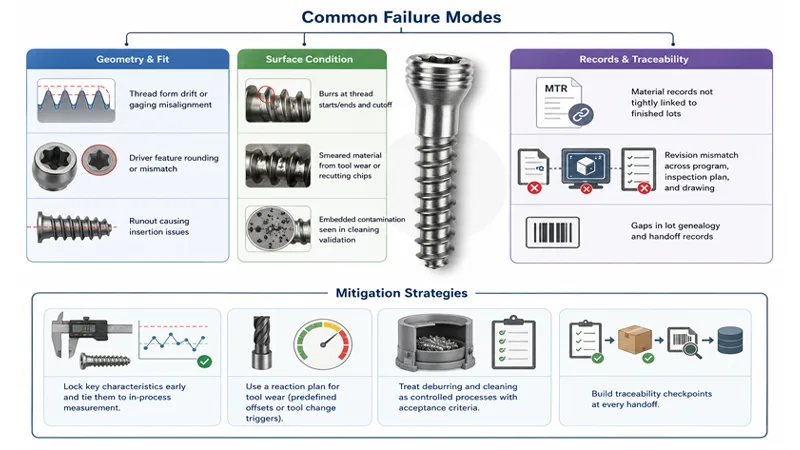
Common failure modes in titanium bone screws turning usually map to three buckets: geometry, surface condition, and records.
Geometry and fit:
- Thread form drift or poor functional gaging alignment
- Driver feature rounding or dimensional mismatch
- Runout that shows up as insertion issues
Surface condition:
- Burrs at thread starts/ends and cutoff
- Smeared material from tool wear or recutting chips
- Embedded contamination that shows up in cleaning validation
Records and traceability:
- MTR not tightly tied to finished lots
- Revision-control mismatch between programming, inspection plan, and drawing
Mitigation strategies that typically work:
- Lock key characteristics early and tie them to in-process measurement.
- Use a reaction plan for tool wear (predefined offsets or tool change triggers).
- Treat deburring and cleaning as controlled processes with acceptance criteria.
- Build traceability checkpoints at every handoff.
Hidden cost drivers and quotes
Quotes for bone screws can look simple (piece price + lead time) but the real cost often includes:
- Tooling strategy changes (thread whirling heads, form tools, custom gages)
- Inspection burden (thread measurement method, driver gages, CMM time)
- Deburring and cleaning steps that weren’t scoped
- Packaging requirements (sterile barrier components, sealing validation, label control)
- Change-control cost when drawings evolve during NPI
When comparing suppliers, insist on a quote that calls out:
- What is included in inspection deliverables (FAI format, sampling plan)
- Assumptions about bar stock condition and certification
- Any outside processes and how traceability is maintained
A clean quote is not the cheapest quote. It is the one that reduces surprise rework and schedule risk.
Prototype-to-ramp scalability plan
A credible prototype-to-ramp plan shows how the supplier transitions from “making it once” to “making it the same way every time.”
What to look for:
- Prototype phase: process proof, gage strategy, and initial capability signals
- Verification phase: tightened controls, stabilized tooling, controlled documentation set
- Production ramp: capacity planning, backup tooling, and defined change control
Ask directly: what changes between prototype and production? If the answer is “nothing,” be skeptical. A good supplier can name the changes and explain why they reduce risk.
Conclusion
Key takeaways for evaluating titanium bone screws turning suppliers:
- Grade 23 (Ti-6Al-4V ELI) raises expectations on incoming material control and traceability.
- DFM decisions around threads and driver features determine downstream burr risk, inspection complexity, and yield.
- Process control is the differentiator: documented parameters, chip/burr strategy, in-process measurement, and SPC with a real reaction plan.
- Cleanliness and packaging readiness should be planned from day one, using standards language (ISO 19227 and ISO 11607) to align stakeholders.
- Regulatory readiness is mostly documentation discipline: revision control, FAI-style evidence, and traceability through every handoff.
Next steps to de-risk sourcing and accelerate timelines:
- Share your drawing pack and size matrix, then request a DFM review focused on thread strategy, driver feature manufacturability, and an inspection plan aligned to your critical-to-quality (CTQ) list.
- Ask for a sample documentation bundle (MTR traceability example + in-process SPC snapshot + FAI-style report) before you commit to a ramp schedule.
FAQ
Titanium Grade 23, also known as Ti-6Al-4V ELI (Extra Low Interstitials), is preferred because it offers superior ductility and fracture toughness compared to standard Grade 5. In the context of Titanium Grade 23 bone screw CNC turning, the lower oxygen and iron content make the material more reliable under the high-stress, cyclic loading conditions typical of orthopedic implants.
Traditional single-point threading can struggle with the heat and toughness of Grade 23 titanium. Thread whirling uses high-speed rotating cutters to “whirl” around the bar stock, producing the entire thread form in one pass. This method is a staple of high-quality Titanium Grade 23 bone screw CNC turning because it significantly reduces burr formation, manages heat better, and ensures a more consistent thread profile across thousands of parts.
While both are titanium alloys, Grade 23 (ELI – Extra Low Interstitial) has stricter limits on elements like oxygen, nitrogen, and iron. This “purity” significantly improves ductility and fracture toughness, which are critical for orthopedic implants that must endure high fatigue loads within the human body without failing.
The driver feature (whether it’s a Hex, Torx-style, or custom lobe) is often where programs face the most risk. During Titanium Grade 23 bone screw CNC turning, sharp internal corners in a driver pocket can act as stress risers or traps for machining debris. We recommend specific radius values and depth-to-width ratios to ensure the driver is both manufacturable and strong enough to withstand high torque without stripping in the OR.
In the medical world, the paperwork is as important as the part. For every Titanium Grade 23 bone screw CNC turning project, we provide a complete Material Test Report (MTR) that traces the finished screw back to the specific “heat” of the raw titanium bar. We use a strict traveler system to ensure that different lots never mix and that all processing—from turning to passivation—is fully documented for your Device History Record (DHR).
Machining residues like oils and metallic fines must be completely removed to ensure biocompatibility. Following Titanium Grade 23 bone screw CNC turning, parts typically undergo a validated cleaning process aligned with ISO 19227. This often involves ultrasonic cleaning and a passivation step (to enhance the protective oxide layer) to ensure the screws are ready for terminal sterilization and won’t trigger an adverse biological reaction.


