In the realm of medical device manufacturing, “biocompatible machining” is not merely a production technique; it is a critical control point that directly impacts patient safety and surgical success. As a senior engineer at AFI Parts, I often emphasize to our clients that machining implants from PEEK (Polyetheretherketone) and Titanium requires a mindset shift from “making parts to print” to “manufacturing life-sustaining devices.”
Unlike standard industrial or automotive components, medical implants fall under strict regulatory scrutiny (including ISO 13485:2016 and FDA 21 CFR Part 820). The process involves navigating complex material behaviors—such as the thermal sensitivity of semi-crystalline polymers and the rapid work-hardening characteristics of reactive metals—while ensuring the finished product remains chemically inert, structurally sound, and biologically safe. This comprehensive guide details the rigorous engineering protocols, scientifically validated processes, and unyielding quality standards we employ at AFI Parts to handle PEEK and Titanium composites, offering actionable insights for product designers, biomechanical engineers, and quality control (QC) specialists.
Table of Contents
Regulatory Challenges Facing PEEK Polymer Application in Medical Devices
Navigating the complex and ever-evolving regulatory landscape is the fundamental first step in biocompatible machining. It is entirely insufficient to simply source “medical-grade” raw material; the CNC machining process itself must be proven not alter or degrade the material’s intrinsic biocompatibility profile. Any unvalidated process step introduces the risk of cytotoxicity, systemic toxicity, or long-term rejection in the patient’s body.
The Landscape of Compliance
You must seamlessly align your precision manufacturing process with stringent global standards. At AFI Parts, our comprehensive process validation architecture is built systematically around the following international frameworks:
- United States (FDA): Compliance with 21 CFR Part 820 (Quality System Regulation) is non-negotiable. Specifically, Subpart G (Production and Process Controls) mandates that any process whose results cannot be fully verified by subsequent inspection must be validated with a high degree of assurance. For Class II and Class III devices (e.g., spinal cages, pedicle screws, and cranial plates), we actively support our customers in their 510(k) or PMA submissions by providing full, uncompromised material traceability (Chain of Custody) and exhaustive Device Master Record (DMR) support.
- European Union (EU MDR 2017/745): The updated Medical Device Regulation requires exceptionally strict technical documentation and clinical evaluation. As a custom metal parts manufacturer, we must scientifically prove that our machining process (e.g., the precise selection and application of cutting coolants, tapping fluids, and anti-rust oils) does not leave microscopic residues that compromise the biocompatibility safety limits established by the ISO 10993 series..
- Japan (PMDA) & China (NMPA): Both regulatory bodies require rigorous physical property testing and biological evaluation of the final machined component, not just the raw stock material. This includes accelerated aging tests and simulated anatomical loading.
Key Regulatory Requirements Table
The following expanded table summarizes the critical regulatory checkpoints we actively integrate into our manufacturing flow to ensure zero-defect medical machining:
| Region | Regulatory Body | Key Standard | AFI Parts Compliance & Validation Focus |
| United States | FDA | 21 CFR Part 820 | Strict control over imported raw materials, vendor qualification, and cleanroom manufacturing environment. |
| EU | MDR | ISO 13485:2016 | Advanced Risk Management (ISO 14971) specifically regarding process-induced contamination and failure modes. |
| Japan | PMDA | JIS T 0993-1 | Sub-micron surface residue analysis, foreign matter exclusion, and heavy metal limit verification. |
| China | NMPA | GB/T 16886 | Strict control over imported raw materials and manufacturing environment, mirroring international biological evaluation protocols. |
| Global | ISO | ISO 10993 | Biological evaluation of medical devices—ensuring our specialized machining fluids don’t cause cytotoxicity or skin sensitization. |
Engineering Insight: You must conclusively validate that your post-machining cleaning process (e.g., chemical passivation or multi-frequency ultrasonic cleaning) effectively removes manufacturing aids to a microscopic level that entirely satisfies ISO 19227 (Cleanliness of Orthopedic Implants—General Requirements). Total Organic Carbon (TOC) thresholds are typically restricted to < 0.5 mg/part.

Understanding PEEK and Titanium as Biomaterials
To custom machine these advanced materials effectively and repeatedly, a manufacturing engineer must deeply understand their polymer microstructure and metallurgical behavior under extreme dynamic stress.
PEEK Properties for Medical Implants
Polyetheretherketone (PEEK), specifically the implantable grades strictly complying with ASTM F2026, has emerged as the gold standard for radiolucent, load-bearing orthopedic implants. Its unique aryl-ketone-ether molecular backbone provides exceptional chemical resistance and mechanical stability.
- Biocompatibility & Modulus Matching: PEEK exhibits a Young’s Modulus ranging from approximately 3.6 GPa (pure, unfilled grade) up to 18 GPa (carbon-fiber-reinforced grades), which astonishingly closely mimics the modulus of human cortical bone (approx. 14 GPa). This mechanical similarity significantly reduces stress shielding—a detrimental biomechanical phenomenon where excessively rigid metal implants carry too much physiological load, causing the surrounding, under-stressed bone tissue to degenerate and resorb (osteopenia).
- Radiolucency: Unlike metallic alternatives, PEEK does not generate scattering artifacts in X-ray, MRI, or CT scans, allowing orthopedic surgeons to monitor osteogenesis and bone fusion post-surgery clearly and accurately.
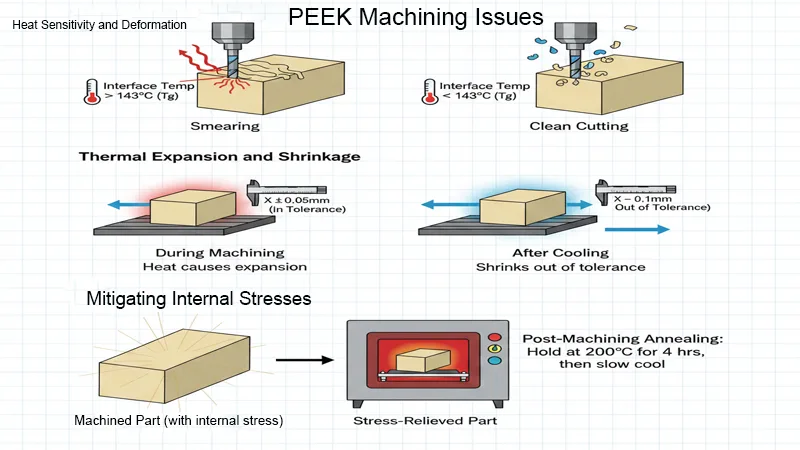
- Thermal Stability and Crystallinity: While PEEK possesses a high melting point (Tm) of ~343°C, its glass transition temperature (Tg) is much lower, situated around 143°C.
- Critical Note: Machining temperatures in the cutting zone that exceed the Tg can induce severe internal residual stresses or fundamentally alter the polymer’s crystallinity (shifting the delicate amorphous vs. semi-crystalline ratio, which should optimally sit around 30-35%), thereby permanently degrading the part’s tensile strength and chemical resistance in vivo.
Titanium and Its Alloys in Implants
For metallic implants requiring ultimate fatigue strength, we primarily utilize Ti-6Al-4V ELI (Grade 23) per ASTM F136 specifications due to its vastly superior fracture toughness and ductility compared to standard commercial Grade 5. ELI stands for Extra Low Interstitials, meaning elements like Oxygen, Nitrogen, and Carbon are strictly minimized during the vacuum arc remelting process.
- Osseointegration: Titanium’s spontaneous formation of an inert, passivating oxide layer (TiO₂) upon exposure to oxygen allows for direct structural, mechanical, and functional connection with living bone—a process vital for implant stability.
- Strength-to-Weight Ratio: With a measured density of ~4.43 g/cm³, titanium is roughly half the weight of traditional medical stainless steel (316L) but offers significantly higher structural integrity, boasting an approximate 860 MPa yield strength.
- Corrosion Resistance: It is virtually immune to galvanic and crevice corrosion in harsh saline body fluids, completely preventing the localized release of toxic metal ions that could trigger metallosis and subsequent tissue necrosis.
Material Comparison for Machinists
| Material | Applicable Standard | Machining characteristic | Biocompatibility Risk |
| PEEK (Unfilled) | ASTM F2026 | Low thermal conductivity (0.25 W/m·K); prone to thermal “smearing,” plastic deformation, and ductile burrs. | Plastic embedding of foreign chips; thermal degradation of polymer chains. |
| CFR-PEEK (Carbon Fiber) | ASTM F2026 | Highly abrasive to cutting tools; causes rapid flank and crater wear on carbide. | Microscopic carbon fiber fallout; inter-laminar delamination exposing porous structures. |
| Ti-6Al-4V ELI (Gr 23) | ASTM F136 | Low thermal conductivity (6.7 W/m·K), severe work hardening, high reactivity (“gummy” nature). | Built-up edge (BUE) galling; localized heat-affected zones and brittle Alpha Case formation. |
Biocompatible Machining Challenges
Precision CNC machining for medical device applications differs fundamentally from aerospace or automotive machining paradigms because, in this sector, “surface integrity” equates directly to “biological safety”. An invisible micro-crack or a microscopic smear of foreign metal can lead to catastrophic clinical failure. Here are the specific, scientifically documented failure modes we guard against continuously at AFI Parts.
PEEK Machining Issues
1. Heat Accumulation and Crystallinity Changes
PEEK acts as a potent thermal insulator rather than a conductor. Unlike conductive metals, it absolutely does not conduct the intense heat of deformation away from the cutting zone via the evacuated chips.
- The Problem: If the tool-workpiece interface temperature rises uncontrollably due to friction, PEEK will undergo localized phase transitions, melting, or surface “smearing”. More critically, excessive heat input can permanently alter the delicate crystallinity percentage of the machined surface layer. A surface that is driven to become too amorphous may be chemically less resistant to harsh sterilization processes (like autoclave or gamma radiation) and biological enzymes.
- The Solution: We rigorously employ “peck drilling” cycles with custom retract planes and programmed interrupted cutting paths to allow critical heat dissipation. We strictly control coolant usage and velocity (see “Techniques” below).
2. Dimensional Instability (Stress Relaxation)
Extruded or molded PEEK stock is highly prone to the release of large residual stresses upon material removal. Removing heavy amounts of material asymmetrically from one side of a billet can cause the part to violently bow, warp, or twist (a defect colloquially known as “potato chipping”).
- AFI Parts Strategy: To combat this, we enforce a mandatory, validated multi-stage machining sequence: Roughing → Annealing (a controlled stress relief heat treatment in programmable ovens) → Precision Finishing. A typical annealing cycle might involve slowly ramping up to 200°C, holding for 4-6 hours based on cross-section, and cooling at a controlled rate of <10°C per hour to prevent thermal shock.
Titanium Machining Issues
1. Work Hardening and Alpha Case Formation
Titanium alloys dynamically work-harden almost instantly if the cutting tool edge rubs rather than cleanly shears the material.
- The Risk: A dull tool, improper rake angle, or critically insufficient feed rate creates a super-hardened, impenetrable surface layer. If extremely high heat is generated in an oxygen-rich environment, it can force the formation of a brittle, oxygen-enriched microstructural layer known as “Alpha Case.” This layer is highly susceptible to micro-cracking and catastrophic propagation under continuous fatigue loading inside the human body.
- Constraint: You simply cannot dwell in a cut. The cutting tool must be aggressively moving, engaged, and shearing at all times to stay ahead of the work-hardened zone.
2. The “Gummy” Nature and Built-Up Edge (BUE)
At elevated machining temperatures, titanium exhibits high chemical reactivity and tends to literally pressure-weld itself to the carbide cutting tool (forming a Built-Up Edge). This alters the tool geometry mid-cut, leading to exceptionally poor surface finishes (tearing) and sudden, unpredictable tool failure.
- Mitigation: This physical phenomenon strictly requires high-pressure coolant (HPC) directed accurately and specifically at the cutting edge to physically shatter the thermal vapor barrier and prevent chemical adhesion.
3. Cross-Contamination Risks
This is perhaps the most insidious hidden danger in multi-material machine shops.
Scenario: A CNC milling center that previously cut naval brass, copper, or leaded free-machining steel absolutely cannot be used for medical-grade PEEK or Titanium implants without executing a fully documented and biochemically validated decontamination protocol. Traces of heavy metals like copper (which is highly cytotoxic) or lead microscopically embedded into the surface of a porous PEEK implant can cause devastating, irreversible biocompatibility failure during ISO 10993 testing.
AFI Parts Standard: To unequivocally eliminate this variable, we utilize strictly isolated, dedicated machine cells exclusively reserved for medical polymers and titanium alloys..

Preparation & Setup for Biocompatible Machining
Before a single chip is cut from the raw material, the entire manufacturing environment must be forensically secured and verified. This section outlines our mandatory “Pre-Flight” engineering checklist for critical medical production.

Workpiece Preparation and Cleanliness
- Material Sourcing: We only source premium medical alloys and polymers from audited, ISO 13485 certified raw material suppliers. Every single bar or billet must arrive with an unimpeachable Material Certification (Mill Cert) verifying full chemical and mechanical compliance with ASTM F2026 (PEEK) or ASTM F136 (Ti-6Al-4V ELI).
- Raw Material Inspection: Upon receipt, we proactively perform FTIR (Fourier Transform Infrared Spectroscopy) on incoming PEEK batches to verify the exact molecular chemical composition and absolutely prevent counterfeit or degraded material entry into our supply chain.
- Handling: Machine operators and inspectors must wear powder-free nitrile gloves at all times. Human skin naturally secretes oils containing complex lipids, squalene, and organic acids that are extremely difficult to completely remove from slightly porous or textured plastics and metals before sterilization.
Cleaning Protocols for Medical Use
Standard industrial shop floor degreasers (like brake cleaner or basic alkaline soaps) are strictly forbidden. We exclusively utilize specific, scientifically validated biocompatible cleaning agents.
- Rough Clean: Initial removal of bulk machining coolant and heavy swarf using filtered deionized (DI) water and medical-grade, free-rinsing mild detergents (e.g., Alconox or Tergazyme).
- Ultrasonic Cleaning: This step is mechanically essential for safely removing microscopic machining fines, embedded particulate, and cutting fluids from complex geometries like deep blind holes, undercut pockets, and fine thread valleys using cavitation.
- Water Quality: Final cascading rinses must be executed with ultra-pure RO/DI water (Reverse Osmosis/Deionized, typically rated at 18 MΩ·cm resistivity) to guarantee the prevention of inorganic mineral deposits (such as calcium or magnesium scales) that could drastically interfere with subsequent passivation layers or final steam sterilization.
Tool Selection and Maintenance
No standard HSS (High-Speed Steel) tooling is permitted in these cells. We strictly standardize on:
- Solid Carbide: Premium sub-micrograin cemented carbide end mills and drills for PEEK to ensure a razor-sharp, wear-resistant cutting edge that shears rather than plows the polymer.
- Polished Flutes: For Titanium alloys, highly polished flutes (and advanced PVD coatings like TiAlN) drastically reduce sliding chip friction and prevent the onset of BUE formation.
- Dedicated Tooling: Cutting tools that have been used on commercial aluminum, brass, or carbon steel are permanently quarantined and never rotated onto the medical production machines. Cross-contamination from uncontrolled tool transfer is a highly documented, catastrophic failure mode in in-vitro cytotoxicity assays.
Equipment and Environment
- Machine Isolation: We deploy “clean” CNC machines running entirely oil-free systems or utilizing strictly medical-approved, water-soluble coolants (typically advanced vegetable-ester based fluids that are certified non-cytotoxic).
- Coolant Management: Coolant concentration (Brix percentage) and pH levels are metrologically monitored and recorded daily. Uncontrolled bacterial or fungal growth in stagnant, old coolant sumps can introduce massive colonies of endotoxins (pyrogens) directly onto the surface of the implant, which standard hospital autoclave sterilization cycles may completely fail to destroy or remove.
Machining Techniques for PEEK and Titanium
Achieving medical-grade precision—where geometric tolerances often tighten to an unforgiving ±0.005mm to ensure perfect anatomical fit—requires mastery of cutting physics. This section details the specific operational parameters and CAM (Computer-Aided Manufacturing) strategies we deploy.
Cutting and Milling PEEK
Speed, Feed, and Cooling
Coolant Strategy: While completely dry machining is theoretically possible for PEEK to totally avoid fluid contamination, it poses a massive, unacceptable risk of localized heat buildup and part distortion. We strongly prefer utilizing a targeted dry air blast (through a vortex tube) or ultra-pure Deionized Water mist. We completely avoid all hydrocarbon-based traditional cutting oils, as the PEEK polymer matrix can microscopically absorb them over extended cycle times, leading to biocompatibility failure.
Cutting Speeds (SFM):
- Unfilled PEEK: Maintain aggressive speeds of 400 – 600 SFM (Surface Feet per Minute) to ensure clean shearing.
- Carbon-Filled PEEK: Drastically reduce speeds to 300 – 400 SFM due to the extreme abrasiveness of the chopped carbon fibers, which act like microscopic grinding wheels against the carbide edge.
Chip Load: Maintain a relatively high and consistent chip load (typically 0.004″ – 0.008″ per tooth for heavy roughing passes). If the programmed feed rate is too slow, the tool edge rubs the plastic, generates massive friction heat, and instantaneously melts the PEEK, destroying the part’s surface integrity.
Tool Geometry
High Positive Rake: Utilizing a rake angle of 12° to 18° effectively slices and shears the plastic cleanly, rather than plowing it and generating compressive heat.
Number of Flutes: Use strictly 1 or 2 flute configurations for end mills. This specialized geometry provides an exceptionally large flute gullet for rapid, unhindered chip evacuation. PEEK chips tend to be continuous, hot, and highly stringy; attempting to use standard 4-flute or 5-flute tools will cause the cutter to clog instantly, leading to catastrophic friction and total part melting.
Machining Titanium Alloys
For complex geometric titanium machining, understanding the Material Removal Rate (MRR) dynamics is essential. The process must be mathematically optimized.
Lubrication and Cooling
High-Pressure Coolant (HPC): We universally utilize advanced 1000 PSI (70 bar) through-spindle coolant (TSC) systems. This massive hydraulic force physically blasts hardened chips out of deep blind pockets and aggressively forces the liquid coolant directly between the tool flank and the workpiece face to prevent destructive heat generation.
Supercritical CO2 (scCO2): For extreme precision components (like micro-vascular clips), we actively experiment with advanced scCO₂ cooling systems, which eliminates thermal shock and keep the critical cutting zone in a cryogenic state, entirely preventing the dreaded alpha-case microstructural formation.
Minimizing Tool Wear
Strategy: We deploy complex Trochoidal Milling (Dynamic Milling) algorithms generated by advanced CAM software. Instead of heavily engaging the full tool diameter (traditional slotting), we program light, rapid radial cuts (engaging only 5-10% of the tool diameter) at exceptionally high spindle speeds and feed rates. This technique drastically reduces the physical arc of contact, allowing the tool core to thermodynamically cool down during the non-cutting portion of the rotation.
Climb Milling: Machinists must always strictly utilize climb milling (which creates a thick-to-thin chip formation) for Titanium alloys. Utilizing conventional milling forces the cutting edge to enter the cut through the severely work-hardened layer generated by the previous pass, rapidly destroying the cutting edge and inducing severe chatter.
Drilling Deep Holes
- Titanium: We strictly use high-performance, internally cooled solid carbide drills. A single programmed peck cycle should absolutely not exceed 1x the drill diameter in depth to proactively prevent catastrophic chip packing and drill snapping.
- PEEK: Drill bits must be maintained in a razor-sharp state. Dull drills will induce extreme friction, causing the localized PEEK material to thermally expand outward during the cut, and then aggressively shrink inward once the drill is retracted, resulting in an unrecoverable “undersized hole” defect.
Surface Integrity and Biocompatibility
The microscopic topological surface finish (measured in Ra, Sa, Rz) directly determines how human biological cells adhere to and interact with the implanted device.
Achieving Medical-Grade Surface Finish
Bone Contact Surfaces: Implants designed for structural integration often require a highly specific, controlled macro-roughness (typically Ra 1.6 – 3.2 µm) or a complex porous textured surface to actively encourage osteoconduction (the physical ingrowth of bone tissue into the titanium surface). We precisely achieve this topography via controlled, automated bead blasting using medical-grade inert media (like virgin aluminum oxide or glass beads).
Articulating Surfaces: Conversely, components acting as dynamic joints (e.g., femoral heads in hip joints) require absolutely flawless mirror finishes (often Ra < 0.05 µm) to dramatically minimize abrasive friction and long-term wear against their mating Ultra-High Molecular Weight Polyethylene (UHMWPE) cup liners. This extreme surface state is achieved through specialized precision CNC grinding, lapping, and controlled electropolishing.
Polishing and Deburring
Microscopic burrs are clinically unacceptable. In the dynamic environment of the human body, a detached titanium burr instantly becomes a dangerous foreign body that triggers an aggressive macrophage response, localized inflammation, and potential implant loosening.
- Manual Deburring: Highly skilled technicians perform manual deburring under strictly controlled 10x – 40x magnification stereoscopic microscopes using specialized ceramic blades.
- Cryogenic Deburring: For complex PEEK geometries, we utilize automated cryogenic tumbling. Liquid nitrogen flash-freezes the polymer, rendering the thin flash and burrs highly brittle; they cleanly snap off during the tumbling action without dimensionally altering the core part geometry.
- Thermal Deburring: Note that thermal deburring (using explosive gas mixtures) is strictly not suitable for medical PEEK due to the extreme risks of unrecoverable polymer chain degradation and surface melting.
Cleaning and Decontamination
This represents the absolute final barrier to patient safety. We mandate a fully validated, multi-stage automated cleaning line:
Final Rinse: Submersion in 18 MΩ ultrapure deionized water.
Pre-soak: Parts undergo a prolonged soak in a specialized proteolytic enzymatic solution engineered to biologically break down any trace organic residues, lipids, or proteins from handling.
Ultrasonic: Transferred to multi-frequency ultrasonic cascaded tanks (sweeping between 40kHz and 80kHz) to gently but thoroughly dislodge deeply embedded sub-micron particulates.
Passivation (Titanium): Strictly conforming to ASTM A967 or ASTM F86 aerospace and medical standards. We meticulously treat titanium components with controlled baths of Nitric or Citric acid to selectively dissolve and remove trace free iron from the surface and rapidly enhance the thickness of the natural TiO₂ oxide layer, exponentially improving corrosion resistance in vivo.
Post-Machining Protocols
Finished parts are thoroughly dried in dedicated, calibrated HEPA-filtered forced-air ovens to categorically prevent airborne particulate re-contamination. They are subsequently meticulously double-bagged and heat-sealed in ISO Class 7 cleanroom-grade low-density polyethylene (LDPE) medical bags before shipment.
Quality Control and Validation for Medical Implants
At AFI Parts, advanced Quality Control (QC) is never treated as a final-step afterthought; it is deeply integrated into the DNA of every manufacturing step.

Inspection Methods
CMM (Coordinate Measuring Machine): Utilized for highly complex 3D geometric validation, GD&T profiling, and positional tolerance verification to sub-micron accuracy.
OGP / Vision Systems: Non-contact optical metrology measurement is incredibly crucial for inspecting PEEK parts, as the spring-loaded ruby styli of mechanical CMM probes can easily compress and permanently deform soft, thin-walled plastic features, yielding false readings.
Profilometers: Utilizing advanced stylus contact or laser-interferometer-based systems to rigorously map and verify 2D and 3D surface roughness parameters (Ra, Rz, Sa).
Biocompatibility Testing & Validation
It is an industry maxim that while we meticulously machine the parts, it is the underlying process that is ultimately validated to ensure repeatability. We utilize a rigorous FDA-compliant statistical validation protocol:
- IQ (Installation Qualification): Exhaustively verifying that the new CNC machine, its chillers, and software are installed correctly to the manufacturer’s exact specifications.
- OQ (Operational Qualification): Statistically verifying via fractional factorial experiments that the machine can repeatedly hold extreme tolerances even when pushed to the outermost limits of its defined operation window.
- PQ (Performance Qualification): Executing three consecutive, independent commercial production runs that must yield absolute zero defects, proving long-term process capability and stability (requiring a statistical Process Capability Index Cpk > 1.33).
Documentation and Traceability
We meticulously maintain an unbreakable “Golden Thread” of lot traceability.
- DHR (Device History Record): A massive, comprehensive packet containing the original raw Material Mill Certs, signed Operator Logs, CNC Tooling Logs, oven Heat Treat Charts, chemical Passivation Logs, and the Final Metrology Inspection Report.
- UDI (Unique Device Identification): For regulatory compliance, we utilize ultra-precise laser marking (specifically using a “dark annealing” style for stainless/Titanium that does not ablate material, keeping the surface perfectly smooth) to apply highly legible 2D Data Matrix codes for FDA inventory tracking and recall capability.
- Note: The laser marking focal depth and energy intensity must be flawlessly controlled; excessive ablation can create micro-notches acting as catastrophic fatigue initiation points in load-bearing implants.
Safety and Compliance in Machining Medical Implants
In our facility, “Safety” dictates a dual mandate: protecting the machine operator on the floor, and ensuring the ultimate safety of the end patient in the operating room.
Operator Safety Protocols
Titanium Dust Fire Mitigation: Fine titanium chips, dust, and swarf are highly reactive and present a severe, explosive flammability risk (classified as a Class D metal fire hazard under NFPA 484). Specialized, intrinsically safe wet dust collectors are absolutely mandatory in these cells. Dry grinding of titanium without heavy flood coolant or inert noble gas shielding is strictly prohibited under our safety manual.
Coolant Mist Control: High-velocity machining generates aerosolized coolant. Advanced mist collectors equipped with multi-stage HEPA filters are installed to protect operators from inhaling airborne bio-hazards or chemical fluids, complying with strict OSHA Permissible Exposure Limits (PEL).
Regulatory Compliance
We rigidly adhere to the dictates of ISO 13485:2016. This systemic commitment means:
- Change Control: A machinist cannot simply change a worn cutting tool brand to a cheaper alternative or alter a coolant concentration type without triggering a formal engineering risk assessment, a review board meeting, and potentially a complete, costly process re-validation.
- CAPA (Corrective and Preventive Action): If a dimensional defect is detected during QC, we don’t just casually fix the part or scrap it; we are required by law to scientifically investigate and document the definitive root cause (e.g., asking “Why did the 1mm drill wander off-center?” -> Utilizing dial indicators to trace the root cause back to “Microscopic spindle runout drift caused by bearing wear”).
Environmental Controls
Our dedicated medical machining cells operate relentlessly under strictly monitored, cleanroom-adjacent controlled conditions.
- Temperature & Humidity: The ambient air is statically maintained at a perfect 20°C ± 1°C.
- Dimensional Impact: PEEK possesses a notoriously high Coefficient of Thermal Expansion (CTE). Machining a part rapidly at an elevated ambient of 25°C and later officially inspecting it on a CMM in a 20°C metrology lab will physically shrink the geometry, directly leading to the catastrophic rejection of highly expensive, tight-tolerance parts.
- Particulates: Hospital-grade positive pressure air filtration drastically reduces microscopic dust that could silently settle onto vulnerable implants during the machining cycle.
Summary
Precision CNC machining of PEEK and Titanium composites for life-critical medical implants is an incredibly demanding discipline that seamlessly merges advanced metallurgy, complex polymer science, fluid dynamics, and unyielding regulatory adherence. It strictly requires state-of-the-art robust equipment, extensively validated engineering processes, and an uncompromising corporate culture dedicated entirely to quality.
At AFI Parts, as a premier custom metal parts manufacturer, we fundamentally understand that behind every CAD drawing and blueprint is a living patient. By strictly adhering to current ASTM biomaterial standards, aggressively utilizing our dedicated biocompatible machining cells, and systematically maintaining rigorous ISO 13485 quality management systems, we proudly deliver flawless components that design engineers trust implicitly and orthopedic surgeons rely on daily.
Ready to discuss your next breakthrough medical device project or optimize an existing product? Contact our senior engineering team today for a comprehensive Design-for-Manufacturability (DFM) review.
FAQ
Biocompatible machining is a highly regulated, statistically validated manufacturing process that mathematically and scientifically ensures the final machined medical device retains the exact biological safety profile and inertness of the original raw material. It involves exhaustive, documented control over coolants, tooling materials, machine environments, surface integrity metrics, and rigorous cross-contamination prevention to pass stringent ISO 10993 cytotoxicity standards.
This is mandated to completely prevent catastrophic metallic cross-contamination. If a carbide end-mill previously used to cut heavy carbon steel is subsequently used to cut PEEK, microscopic iron or heavy metal particles can physically shear off and embed deeply into the porous plastic matrix. Later, in an MRI machine, these embedded ferrous particles can inductively heat up severely or create massive image artifacts, blinding the radiologist. Inside the body, they can cause severe tissue necrosis or inflammation.
Mere visual inspection is useless for medical implants. Surface cleanliness is rigorously validated through Total Organic Carbon (TOC) testing, Gravimetric analysis (microscopic particle counting), and sensitive in-vitro Cytotoxicity testing to definitively ensure absolutely no trace manufacturing residues (like hydrocarbon oils, synthetic detergents, or bacterial endotoxins) remain on the part.
It heavily depends on the specific anatomical function. For areas requiring osseointegration (structural bone growth into the implant), a controlled macro-roughness of Ra 1.0 – 2.0 µm is highly targeted to give osteoblasts a scaffold to grip. Conversely, for articular, moving surfaces (like rubbing knee joints), an ultra-polished, mirror-like finish of Ra < 0.1 µm is strictly required to drastically minimize kinetic friction and particulate wear.
Generally, absolutely no. To pass FDA clearance, water-soluble, highly refined vegetable-ester based, or synthetic coolants that are laboratory-certified as non-cytotoxic and completely free of heavy metals, sulfurs, and bactericides are strictly required. To entirely eliminate this immense risk variable, many advanced medical machine shops opt for pure dry machining (with cold air guns) or lubricate strictly with highly filtered pure water.
You must strictly maintain legally auditable “cradle-to-grave” traceability. This unbreakable chain perfectly links the specific 2D laser-etched implant serial number back through the entire production history to the exact CNC machine utilized, the logged operator that day, the specific ultrasonic cleaning batch, and finally back to the original titanium smelter’s raw material melt/lot number.


